By Saadia Aslam
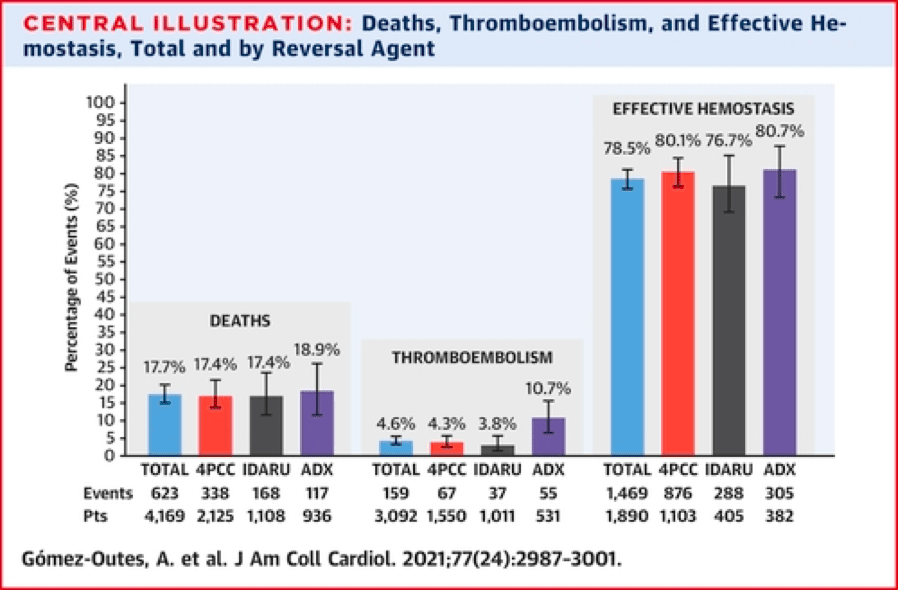
Major bleeding that requires hospitalisation occurs in 2% to 3.5% of patients that take DOACs. Some of these patients may require reversal agents to control bleeding. Recent work from Gómez-Outes et al evaluated clinical outcomes associated with the use of 4-factor prothrombin complex concentrates, idarucizumab, or andexanet in this setting.This evaluation included 60 studies in 4,735 patients with severe DOAC-related bleeding who were treated with 4-factor prothrombin complex concentrates (n = 2,688), idarucizumab (n = 1,111), or andexanet (n = 936).
- Mortality rate: 17.7% (95% confidence interval [CI]: 15.1% to 20.4%), and this was increased in patients with intracranial bleedings (20.2%) than in patients with extracranial haemorrhages (15.4%).
- Thromboembolism rate: 4.6% (95% CI: 3.3% to 6.0%), which was particularly high with andexanet (10.7%; 95% CI: 6.5% to 15.7%).
- Effective haemostasis rate: 78.5% (95% CI: 75.1% to 81.8%) and was similar amongst the reversal agents.
- Re-bleeding rate: 13.2% (95% CI: 5.5% to 23.1%) of which 78% re-occurred after resumption of anticoagulation.
- Risk of death: markedly and significantly associated with failure to achieve effective hemostasis (relative risk: 3.63; 95% CI: 2.56 to 5.16).
This has highlighted that DOAC-related bleeding is associated with high rate of mortality despite effective haemostasis. Furthermore, thromboembolic rates are particularly high with andexanet. Comparison trials between different reversal agents are needed.
Reference:
Gómez-Outes A, Alcubilla P, Calvo-Rojas G, et al, Meta-Analysis of Reversal Agents for Severe Bleeding Associated With Direct Oral Anticoagulants. J Am Coll Cardiol 2021;77:2987-3001.
Read more:
https://www.jacc.org/doi/10.1016/j.jacc.2021.04.061?_ga=2.179849892.1247377718.1624477281-193636431.1624373337&
https://www.acc.org/latest-in-cardiology/journal-scans/2021/06/15/14/00/meta-analysis-of-reversal-agents